Capabilities
Diligent Accord maintains state-of-the-art medical manufacturing facilities and operates against stringent standard operating procedures (SOPs), quality checks and traceability requirements required by our customers and medical certification.
Track Record
Supported by decades of healthcare manufacturing experience for Tier 1 OEMs, Diligent Accord has a proven track record in the production of quality-critical medical products, delivered on-time straight to the door of our customers.
Certification
Diligent Accord maintains several industry certifications critical to the needs of the global healthcare manufacturing industry, including ISO:13485 for medical devices, FDA certification for various medical products and finished goods and ISO:15001 compliant facilities for ultrasonic cleaning.
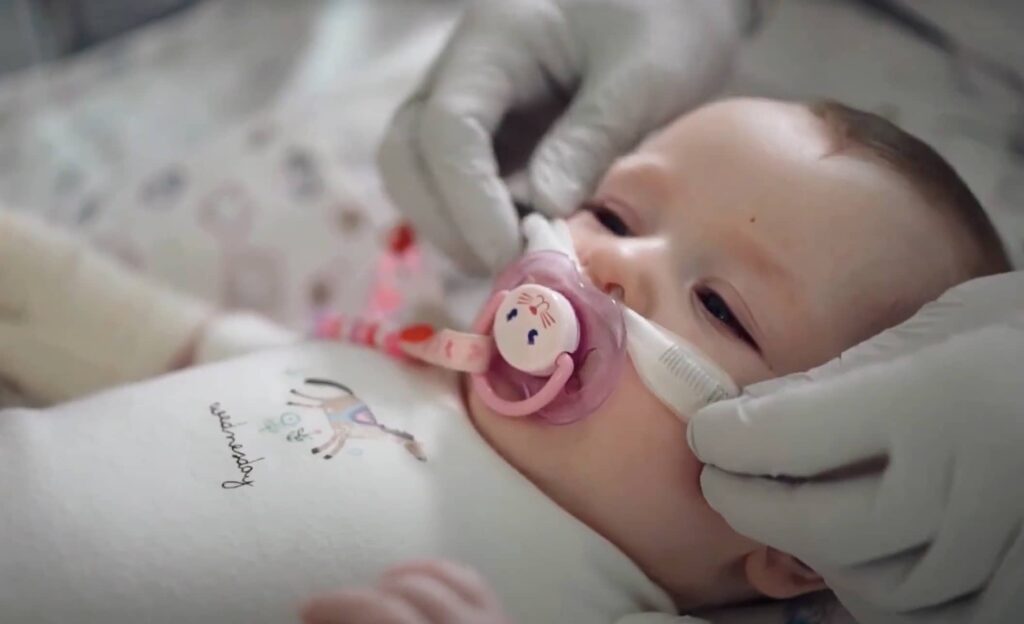
Diligent Accord maintains a flagship focus on serving the global healthcare & medical devices industry across the design, manufacturing and assembly of complex, quality-centric parts and products. This is supported by decades experience and multiple tier 1 OEM healthcare customers, many having been with Diligent Accord for more than a decade.
Diligent Accord’s in-house capabilities, machinery, facilities and suite of industry leading certifications are expertly aligned to the needs of the global healthcare industry and our customers. Diligent Accord employs a number of healthcare SMEs across its production facilities, business and QA functions, providing critical industry-specific insight.
Diligent Accord maintains a number of purpose-built facilities to serve its healthcare client base, including dedicated medical and ultrasonic cleaning facilities and secluded, ISO:15001 compliant controlled working environments. Diligent Accord also operates a series of standard operating procedures (SOPs) and quality checks aligned with the medical industry, and in many cases creates customised processes to align with the documentation used or required by the customer.


Diligent Accord’s suite of industry certifications enables it to manufacture almost any medical product, to any specification. This extends, but is not limited to:
This certification has allowed Diligent Accord to design, manufacture and assemble millions of high-value medical products to date including complex medical insufflators, healthcare consumables, surgical equipment, breathing apparatuses and much, much more.
We operate on a project-by-project basis, and work collaboratively with our clients on all design engagements. All project files, scope documents and design details are available online, providing customers with a ‘live’ feed to monitor project progress and comment freely.
We take preliminary concepts from 2D drawings or 3D models through an iterative design process, selecting the appropriate design software and project management methodology to drive the engagement.
All products must then go through a rigorous testing cycle before the final prototyping and validation phase. Only when the finished product is perfect, all items agreed in our initial scope assessment are complete and all parties are happy can final designs be signed off.
At Diligent Accord, the quality of our products are our greatest priority.